Available 1 January 2023 for:
(a) non-exclusive corporate licensing by individual human health care and veterinary IVD manufacturers;
(b) potential exclusive license for select health care diagnostic fields of use, or non-exclusive license with ROFN;
(c) collaborative research and development agreement, with licensing conversion rights; or
(d) exploratory positioning for partnering within environmental, cosmetics, and food industries
To explore an opportunity regarding Dr. Claudio O. Egalon, Ph.D’s invention, please contact Bio-Investigations Ltd. (bioinv1@aol.com).
Inquiries will be held in confidence.
—
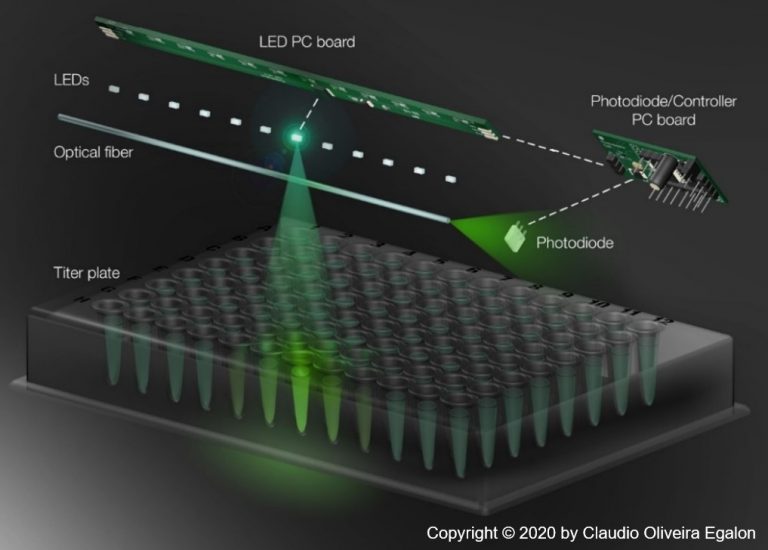
Science and Sensors Technologies (S&ST) has invented, designed, manufactured, and tested a Titer Plate Reader interface (TPRi), utilizing Side Illuminated Fiber Optics (SIFO). This simple device is being used as an interface between the sample wells of a titer plate and the detection system of existing readers. It simplifies the architecture of TPRs, reduces their costs, and improves their performance. As an example, there are currently available readers that incorporate a matrix of 96 photodiodes, with one for each titer plate sample — by using S&ST’s interface, there is the opportunity to reduce this number of photodiodes to 8, resulting in a 10-fold savings in the detection system. Other benefits exist in terms of performance such as:
- Low cost.
- No moving parts.
- Simple architecture.
- Interchangeable light sources.
- Increased signal and resolution.
- High density of measurement points.
- Light sources of different wavelengths.
- Simultaneous and quasi simultaneous measurements.
- Limited number of detectors: one detector for every 12, or more, wells.
- Reduction in the number of expensive spectrometers that need to be used.
- Use of multiple LEDs to control the signal and resolution of the measurement.
- Leverages the electronics, software, and signal processing capability of existing TPRs.
- Fluorescence, chemiluminescence, colorimetric and scattering measurements, among others, are all applicable.
All these advantages result in several additional benefits:
- Multiple LEDs along the wells of a TPR increase the measurement redundancy.
- Multiple sources provide multiple independent measurements of the same parameter in different wells and increases the TPR measurement precision.
- Multiple independent measurements of different parameters in the wells increases the TPR specificity.
- Multiple LEDs simultaneously probing the same sample increase the signal and improve the measurement sensitivity and resolution of the TPR.
- Quasi simultaneous measurements of different wells are possible by consecutively pulsing each LED while synchronizing them with their respective detectors.
- Simultaneous measurements from different samples are also possible using sources with non-overlapping spectral distribution and a spectrometer.
- “The LED distance can be matched with the spacing between wells in a titer plate. As a result, we can accommodate hundreds, if not thousands, of measurement points: this is particularly useful for microplates with 384 and 1,536 wells.” COE
The technology behind the SIFO titer plate reader is supported by a proprietary IP fence including 64 issued U.S. and international patents, with the most recent patent being US Patent 10,876,960, dated 29 December 2020.
Reference: The global microplate systems market size is projected to reach USD 1,170 million by 2026 from USD 927 million in 2021, at a CAGR of 4.8% from 2021 to 2026. Applications for microplate readers include chemical, biological, drug discovery, environmental, cosmetics, and food, among others. The microplate readers segment is cited as having the highest growth during the forecast period.
For more information, please contact Stewart Rosenberg at (203) 376-7568.
 |
 |
Certain statements in this “Technology Offering” constitute forward-looking statements, are not historical facts, and involve risks and uncertainties that could cause actual results to differ from those expected and projected. Such risks and uncertainties may include, but are not limited to (i) general economic conditions; (ii) conditions specific to the industry; (iii) the company’s ability to develop or acquire new technology or products through licensing, merger
or acquisition and to obtain regulatory approval to commercialize diagnostic, therapeutic or preventative products; (iv) the effectiveness and ultimate market acceptance of any such products; (v) limitations on third party reimbursements with respect to any such products; and (vi) competition. The company does not undertake to update or revise any forward-looking statements contained herein whether as a result of new information, future events or otherwise.

